One of the main questions of our research, is how does the neural code fundamentally alter how we interact with our world and how does our behavior “close the loop” to alter our neural code? When in any environment, we are not passive sensors, but active participates, where we touch, look, and attend to our surroundings. Through these active processes, our nervous systems become dynamic systems which can detect and discriminate objects on rapid timescales. Through our research in the rodent whisker system, we have begun to explore how bottom up mechanisms such as sensory adaptation, and top-down mediated, brain states, alter how we perceive and behave in our world.
Adaptive Sensing. Our decisions in everyday situations are governed by sensory inputs but also, and most importantly, by our experience in a constantly changing sensory environment. How do we adapt to such contextual changes? What is our strategy to deal with a dynamically changing environment? And how do we harness the adaptive strategies to enhance learning?
In our lab we investigate adaptive perception and behavior in the rodent model by using well controlled behavioral tasks in combination with wide field brain imaging with genetically engineered voltage indicators and electrophysiological recordings. Pairing this with theoretical models for predictions and hypothesis testing, the aim of this research is to better understand and quantify sensory processing, learning, perceptual adaptation and cognitive aspects of behavior.
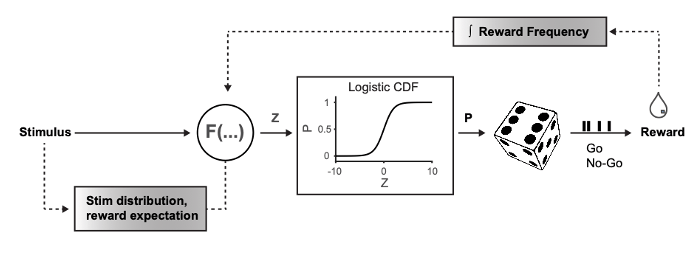
In a recent study we provide evidence that rodent behavioral performance is highly flexible in the face of a changing sensory environment, consistent with a strategy to maintain a desired accumulation of reward. The sensory environment was changed by transforming the probabilistic distribution of whisker deflection amplitudes systematically while measuring the animal’s detection performance and corresponding reward rate. We show that the psychometric function deviates significantly and reversibly depending on the probabilistic distribution of stimuli.
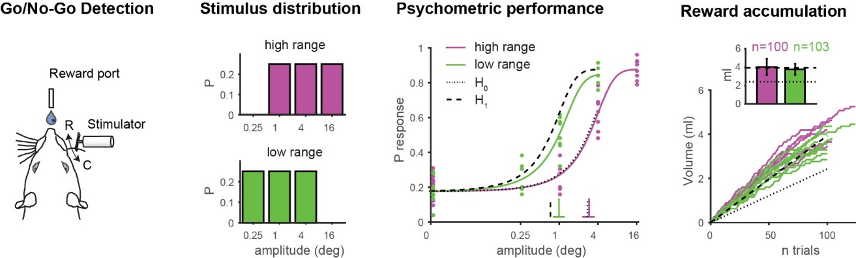 Our simple model of reward accumulation predicts that this change in performance relates to maintaining a constant payoff under changing sensory conditions and
Our simple model of reward accumulation predicts that this change in performance relates to maintaining a constant payoff under changing sensory conditions and
highlights some of the aspects how animals flexibly adjust their behavioral strategy. Furthermore, these findings suggest the existence of an internal probabilistic model that facilitates behavioral adaptation when sensory demands change.
 Future work is underway to determine the role of primary sensory cortex and other downstream structures in the decision making process of this adaptive behavior.
Future work is underway to determine the role of primary sensory cortex and other downstream structures in the decision making process of this adaptive behavior.
C. Waiblinger, C. C. Wu, M.F. Bolus, P. Y. Borden, G. B. Stanley. Stimulus context and reward contingency induce behavioral adaptation in a rodent tactile detection task, J. Neurosci., 39(6):1088-1099, 2019. PDF
Adaptation and Sensory Perception. When we interact with a novel object, our first instinct is to explore the contours and to feel the texture through active sensing. At first we are able to just detect the object, however given repeated stimulation, our sensory system begins to adapt to the stimulus, and we begin to discriminate additional characteristics. This phenomena known as sensory adaptation, leads to an overall decrease in neural activity which fundamentally changes the neural coding properties and serves to improve information transmission of complex inputs. This dichotomy has been heavily examined in our anesthetized work.
Through a number of studies in our lab (and others) using both anesthetized and awake animals, we have shown that the neural code fundamentally changes with sensory adaptation, and more importantly change the behavior of trained animals. Rodents are that can be trained to respond to sensory stimulation of the whisker system, which we have used to validate theories and phenomena shown in anesthetized data sets. In particular, rodents can be trained to detect a whisker deflection, and respond by licking a water spout.

We have shown that through active sensing, such as whisking or repeated whisker movements of 7-12hz, animals reduce their ability to detect sensory information (Ollerenshaw et al 2012). Additionally, we have shown that passively applied whisker adaptation at 12 Hz, can decrease the ability of animals to
detect sensory stimulation while increasing the discriminability of different stimuli (Ollerenshaw et al 2014). Through simultaneous recordings in the thalamus, we have shown that primary sensory thalamic region appears to play a vital role in awake animals in controlling the transmission and neural encoding of sensory information to downstream cortical regions. In particular, the thalamus appears to regulate sensory information by controlling a synchrony code, which can dramatically shift the nonlinearities in the encoding of sensory stimuli.
R. A. Jenks, A. Vaziri, A. S. Boloori, and G. B. Stanley. Self-motion and the shaping of sensory signals, J. Neurophysiol., 103: 2195-2207, 2010. PDF
D. R. Ollerenshaw, B. A. Bari, D. C. Millard, L. E. Orr, Q. Wang, and G. B. Stanley, Detection of tactile inputs in the rat vibrissa pathway, J. Neurophysiol., 108, 479-490, 2012. PDF
D. R. Ollerenshaw, H. J. V. Zheng, Q. Wang, and G. B. Stanley, The adaptive trade-off between detection and discrimination in cortical representations and behavior, Neuron., Mar 5;81(5):1152-64, 2014. PDF
Behavioral and Electrophysiological Effects of Cortical Microstimulation. Electrical microstimulation has been widely used to artificially activate neural circuits on fast time scales.  Despite the ubiquity of its use, little is known about precisely how it activates neural pathways. Current is typically delivered to neural tissue in a manner that provides a locally balanced injection of positive and negative charge,
Despite the ubiquity of its use, little is known about precisely how it activates neural pathways. Current is typically delivered to neural tissue in a manner that provides a locally balanced injection of positive and negative charge,
resulting in negligible net charge delivery to avoid the neurotoxic effects of charge accumulation. Altering waveform shape and using an asymmetric current pulse waveform theoretically reverses this bias and preferentially activates cell bodies, providing increased specificity. We compared the behavioral and electrophysiological effects of symmetric vs. asymmetric current waveform shape in cortical microstimulation.

Using a go/no-go behavioral task, we found that microstimulation waveform shape significantly shifts
psychometric performance, where a larger current pulse was necessary when applying an asymmetric
waveform to elicit the same behavioral response, across a large range of behaviorally relevant current
amplitudes. Using voltage-sensitive dye imaging of cortex in anesthetized animals with simultaneous cortical microstimulation, we found that altering microstimulation waveform shape shifted the cortical activation in a manner that mirrored the behavioral results. Taken together, these results are consistent with the hypothesis that asymmetric stimulation preferentially activates cell bodies, albeit at a higher threshold, as compared to symmetric stimulation.
B. A. Bari, D. R. Ollerenshaw, D. C. Millard, Q. Wang, G. B. Stanley. Behavioral and electrophysiological effects of cortical microstimulation parameters. PLoS One 8: e82170, 2013. PDF



